cnn
—
Here's a look at Alzheimer's disease, a progressive brain disorder that leads to loss of memory and other intellectual abilities.
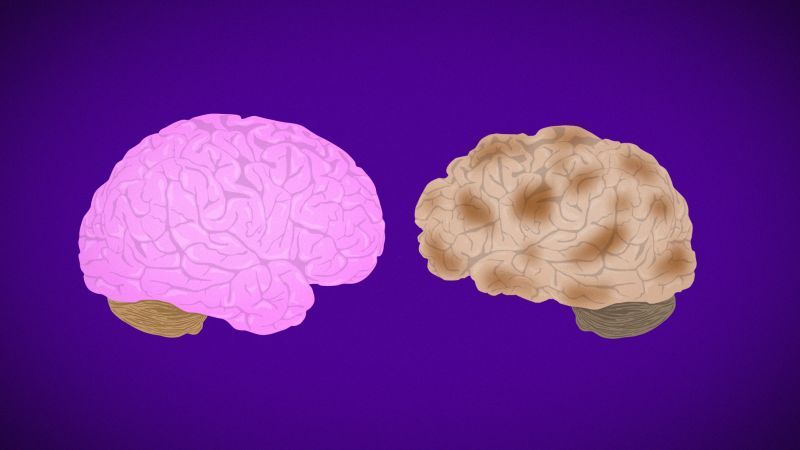
Alzheimer's disease is the most common form of dementia, which is a general term for loss of memory and intellectual abilities.
Alzheimer's disease is fatal and there is no cure. It is a slowly progressive disease that begins with memory loss and ends with severe brain damage.
The disease is named after Dr. Alois Alzheimer. In 1906, the neuropathologist performed an autopsy on the brain of a woman who died after experiencing language problems, unpredictable behavior, and memory loss. Dr. Alzheimer discovered amyloid plaques and neurofibrillary tangles, which are considered hallmarks of the disease.
Contributing factors:
Age – The chance of developing Alzheimer's doubles every five years after age 65. For most people, symptoms first appear after age 60.
Family history – Genetics plays a role in an individual's risk of developing the disease.
head trauma – There is a possible link between the disease and repeated trauma or loss of consciousness.
Heart health – The risk of vascular dementia increases with heart conditions such as high blood pressure, high cholesterol, and diabetes.
Possible symptoms:
Memory loss
Repeated questions and statements.
Lack of criteria
Lost items
Mood and personality changes.
Confusion
Delusions and paranoia
Impulsiveness
Seizures
difficulty to swallow
According to the CDC, an estimated 6.5 million Americans have Alzheimer's disease. It is the fifth leading cause of death among adults in the United States aged 65 and older.
The estimated cost of caring for patients with Alzheimer's or other dementias in the United States in 2023 is $345 billion.
Early-onset Alzheimer's is a rare form of dementia that affects people under 65 years of age.
Early-onset Alzheimer's disease often runs in families.
March 9, 2014 – In a first-of-its-kind study, researchers report that they have developed a blood test that predicts with surprising accuracy whether a healthy person will develop Alzheimer's disease.
September 2014 – The research journal Aging reports that in a small study conducted at UCLA, nine of the 10 patients involved said their symptoms were reversed after participating in a rigorous program that included things like optimizing vitamin D levels in the blood, using supplements of DHA to save connections in the brain and strategic fasting to normalize insulin levels.
September 11, 2015 – The Journal of Neurology publishes a study suggesting that the compound resveratrol, when taken in concentrated doses, may have benefits in slowing the progression of Alzheimer's disease.
November 23, 2016 – American drugmaker Eli Lilly announces that it is completing the Phase 3 clinical trial of its Alzheimer's drug solanezumab. “Patients treated with solanezumab did not experience a statistically significant slowing in cognitive decline compared to patients treated with placebo,” the company says in a statement.
February 2017 – Pharmaceutical company Merck is halting a late-stage trial of its Alzheimer's drug verubecestat after an independent study found it had “virtually no chance” of working.
July 25, 2018 – Additional results from an initial clinical trial of an experimental drug, the antibody called BAN2401, show that it improved cognition and reduced clinical signs of Alzheimer's disease in the brains of study participants. Details about the immunotherapy were announced at a press conference during the 2018 Alzheimer's Association International Conference.
February 28, 2019 – The journal Nature Genetics publishes a study that reveals four new genetic variants that increase the risk of suffering from Alzheimer's disease. The genes appear to work together to control body functions that affect the development of the disease.
March 21, 2019 – Pharmaceutical company Biogen announces that it is completing two Phase 3 clinical trials of a drug that targets the brain's buildup of beta-amyloid, one of two proteins that researchers believe contribute to the development of Alzheimer's. The drug, called aducanumab, was considered a promising candidate for improving cognitive function.
October 22, 2019 – Biogen announces that it will apply for regulatory approval for aducanumab and begin new studies. “The decision to apply is based on a new analysis, conducted by Biogen in consultation with the US Food and Drug Administration (FDA), of a larger data set from Phase 3 clinical studies that were discontinued in March 2019 following a futility analysis.”
March 13, 2021 – The New England Journal of Medicine publishes a study indicating that Eli Lilly and Company's experimental intravenous drug donanemab could slow cognitive decline in patients with Alzheimer's disease, according to early results from clinical trials.
June 7, 2021 – The FDA approves the use of the experimental drug aducanumab for early-stage Alzheimer's disease, even though an FDA advisory committee concluded last year that there is insufficient evidence to support the treatment's effectiveness. This is the first time since 2003 that the FDA has approved a new therapy for Alzheimer's.
April 4, 2022 – A study is published that discovered 42 additional genes related to the development of Alzheimer's disease.
April 7, 2022 – The Centers for Medicare and Medicaid Services announces it will restrict coverage of the controversial and expensive Alzheimer's drug Aduhelm to those enrolled in qualified clinical trials.
May 4, 2022 – The FDA announces that it has allowed the marketing of a new diagnostic test for Alzheimer's disease. This is the first in vitro diagnostic test that could offer an alternative to tools such as PET scanning, which are currently used to diagnose Alzheimer's disease.
June 30, 2022 – Scientists identify a gene that appears to increase the risk of Alzheimer's in women, providing a potential new clue as to why more women than men are diagnosed with the disease. The O6-methylguanine-DNA-methyltransferase gene, or MGMT, plays an important role in how the body repairs DNA damage in both men and women. But the researchers found no association between MGMT and Alzheimer's in men.
January 6, 2023 – The FDA grants accelerated approval to the drug lecanemab, which will be marketed as Leqembi. It has shown “potential” as a treatment for Alzheimer's disease by appearing to slow progression, according to Phase 3 trial results, but has raised safety concerns due to its association with certain serious adverse events, including brain inflammation and bleeding.
July 6, 2023 – The FDA grants traditional full approval to Leqembi, the first drug shown to slow the course of Alzheimer's. The Centers for Medicare and Medicaid Services says it will now expand coverage of the drug, expanding access to an estimated one million people with early forms of the disease.
January 22, 2024 – A new study published in the journal JAMA Neurology suggests that a person's blood test for a type of protein called phosphorylated tau, or p-tau, can be used to detect Alzheimer's disease with “high accuracy,” even earlier. before symptoms begin to appear.